Paper: https://doi.org/10.1117/12.3004498
Highlights
- Novel supervised contrastive pre-training strategy for radiation-induced xerostomia prediction.
- Cluster-guided loss function to improve model performance on imbalanced medical datasets.
- State-of-the-art performance in predicting radiation-induced xerostomia.
- Improved generalization through contrastive learning on limited medical imaging data.
Abstract
Radiation-induced xerostomia is a common side effect of head and neck cancer radiotherapy that significantly impacts patients’ quality of life. Accurate prediction of xerostomia risk before treatment could enable personalized treatment planning. We propose a deep learning framework that combines supervised contrastive pre-training with cluster-guided loss to predict radiation-induced xerostomia. The supervised contrastive learning approach learns robust feature representations from limited medical imaging data, while the cluster-guided loss addresses class imbalance issues common in medical datasets. Our method achieves superior performance compared to existing approaches, demonstrating the effectiveness of combining contrastive learning with specialized loss functions for medical outcome prediction.
Method
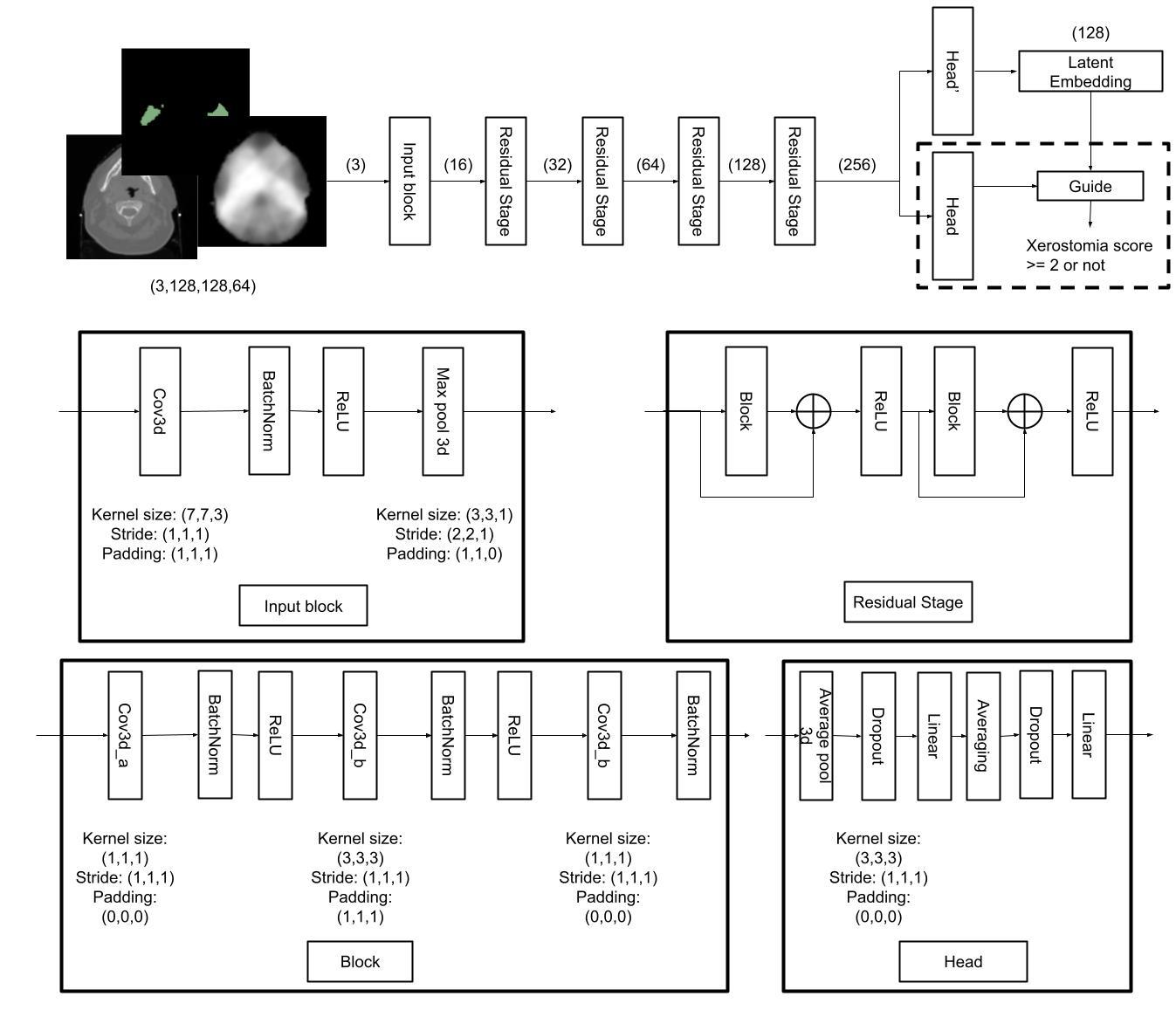
Our method consists of two main components: supervised contrastive pre-training and cluster-guided loss.
The supervised contrastive pre-training phase learns discriminative feature representations by pulling together samples from the same class while pushing apart samples from different classes in the embedding space. This approach is particularly effective for medical imaging tasks where labeled data is limited.
The cluster-guided loss addresses the class imbalance problem by incorporating cluster information into the loss function. This ensures that the model learns to distinguish between different outcome groups even when some classes have significantly fewer samples.
Results
Our framework achieves state-of-the-art performance on xerostomia prediction tasks. The supervised contrastive pre-training significantly improves feature quality, while the cluster-guided loss effectively handles class imbalance. Ablation studies demonstrate the contribution of each component to the overall performance.
Conclusion
This article is only meant for a brief introduction.
We present a novel deep learning framework for predicting radiation-induced xerostomia that combines supervised contrastive pre-training with cluster-guided loss. The supervised contrastive learning approach enables effective learning from limited medical imaging data by learning robust feature representations. The cluster-guided loss addresses class imbalance issues common in medical outcome prediction tasks. Our experimental results demonstrate that this combination significantly improves prediction accuracy compared to existing methods, providing a promising tool for personalized radiation therapy planning.
